The Raine Study supports and facilitates the research community accessing and utilising the enormous potential of this unique cohort to make new discoveries to improve human health.
The principles for researcher engagement with the Raine Study, including the conditions and rules for utilising Raine Study resources and expected behaviour are detailed in the Raine Study Researcher Engagement Policy on our Commonly Used Resources page.
Every new project application goes through a review and approval process that takes on average 6-8 weeks. This includes projects relating to grant/fellowship applications, which must be submitted at least 6 weeks prior to the grant submission deadline. A project application is first reviewed by the Scientific Management Committee (SMC), which meets fortnightly. Following SMC review, the project application is then reviewed by the Scientific Review Committee (SRC), which meets face-to-face once a quarter, with electronic meetings in between as needed.
All projects require formal approval from the Raine Study Scientific Review Committee before the project can commence and there are four key stages in the application process, as outlined below:
For new project applications, we suggest researchers contact the Scientific Management Committee to have an informal discussion about their project idea. The Committee can suggest potential collaborators and advise on relevant considerations, such as data access fees, need for a biosample request, and overlaps with previously approved projects. You can email the Scientific Manager.
Some funding bodies require an expression of interest (EoI) as the first step in a grant/fellowship application. Commonly, an EoI comprises a short summary of the project idea and precedes a full application. Researchers planning to submit a grant/fellowship EoI that proposes the use of Raine Study data should contact the Raine Study Scientific Manager or Scientific Director and discuss the project idea as part of the pre-application process. If the researcher successfully moves into the full grant/fellowship application stage, a project application needs to be submitted in ROSS. The full grant/fellowship application can only be submitted to the funding body after being approved by the Raine Study.
The Lead Investigator will submit a Project application via the Raine Online Submission System (ROSS). On ROSS, the project is only formally submitted after each member of the project team confirms their involvement and acknowledge our code of conduct. Once submitted, the application is reviewed during our fortnightly Scientific Management Committee (SMC) meeting. The SMC will discuss the project and then either place the item on the next Scientific Review Committee (SRC) meeting agenda or contact the Lead Investigator to suggest revisions. The SRC will then discuss the project and either approve, request revisions/amendments or reject it. The SRC meets face to face quarterly and electronically in between the face to face meetings. Please allow a period of 6-8 weeks from the formal submission of the project application to final approval.
Projects can only commence once they have received final approval by the Scientific Review Committee and have all required ethics approvals. Every new project proposing to use existing data and/or biosamples or to collect new data and/or biosamples from the Raine Study participants must have its own Human Research Ethics Committee (HREC) approval from a HREC that is registered with the NHMRC. For more information regarding the ethics approval process for projects using the Raine Study resources please refer to the Commonly Used Resources page and FAQ.
For projects involving the use of existing data and/or biosamples, once final approval by the Scientific Review Committee and from a HREC is obtained, the Lead Investigator will submit a data access request and/or biosamples request via ROSS. On ROSS, the data access request is only formally submitted after each listed data handler confirms their involvement and acknowledge our data access code of conduct. Once submitted, the data access request is reviewed during our fortnightly SMC meeting and the SMC will either approve, request revisions/amendments or reject it. If approved, the Raine Study Data and Biosamples Manager will arrange data extraction and secure transfer to the data handlers. Please note the time for data extraction varies according to the amount and type of data requested.
For projects involving the collection of new data and/or biosamples in Raine Study participants, once final approval by the Scientific Review Committee and from a HREC is obtained, the Lead Investigator will work with the Raine Study team to conduct data collection.
This part of the process has two key steps: manuscript submission and media release/engagement.
In the project application, the research team provides information regarding proposed manuscripts to be completed. For an approved project, once researchers prepare a full manuscript that is almost ready for submission to a peer-reviewed journal, they must submit the draft manuscript to ROSS using the MS Form. The SMC will then review and approve or advise revisions/amendments are required. Following MS approval, the research team can submit the manuscript to a journal.
Please note manuscripts must not be submitted to a journal prior to the Raine Study approval. Manuscripts will be checked for similarity/plagiarism, potential negative impact on the cohort, and appropriate nomenclature and acknowledgements.
If the team wishes to promote their research through the media, they should contact the Raine Study Communications Manager, who will liaise with the research team and their institutional media team as required and approve any media releases related to the Raine Study.
All updates on Raine Study project approvals, rejections and revisions are provided via ROSS through all stages. Any changes researchers would like to make to existing projects need to be made in ROSS as an amendment.
Raine Online Submission System (ROSS)
ROSS has been designed to provide an efficient project management process. Researchers who would like to use data from the Raine Study must submit a project application via ROSS.
Research projects conducted in the Raine Study all progress through four main stages as outlined above. ROSS makes it easier for researchers to provide the information required for their projects to be approved, manuscripts to be reviewed and to manage their involvement in the use of data and/or biosamples.
If you do not have an account yet, please email the Raine Study.
If you already have an account, you can access ROSS here.
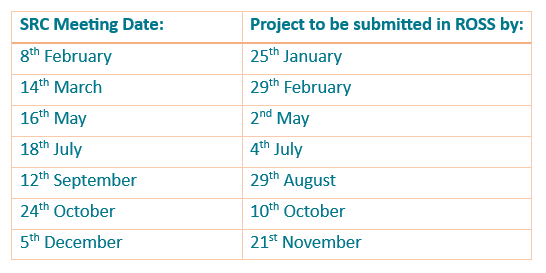
Key dates for 2024
Need more help?
You can find further information about project application, conduct and reporting under our Frequently Asked Questions.
You can also find useful documents, such as Word templates of the forms used in ROSS (that can be used to circulate drafts among co-investigators before completing the final online version in ROSS) under our Commonly Used Resources page.
For help navigating ROSS, please email the Raine Study.
Other Useful Links
For more details regarding the data available from the Raine Study, please check our Available Data page.

